Healthcare System
Economic Benefits of PFC
Post-fracture Care (PFC) coordination programs, such as Fracture Liaison Services (FLS), are the single most important health service intervention to reduce the risk of subsequent fractures. PFC coordination programs are a widely implemented coordinator-based model of care aiming to identify people at risk of subsequent fractures and signposting them to preventive follow-up care services [1]. Numerous studies have demonstrated significant cost-effectiveness through the adoption of PFC coordination programs [2,3,4].

* Dedicated clinical service that finds, tests & treats adults over 50 years with broken bones
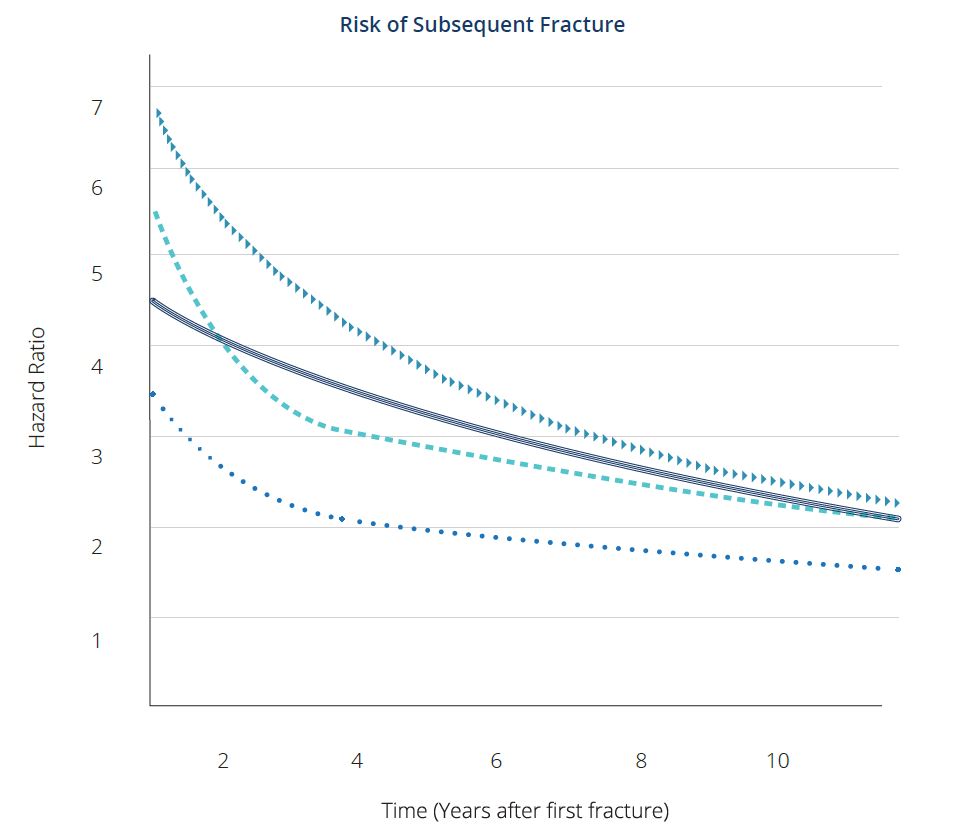
Active research and clinical trials have led to effective treatments. These have been developed, tested and shown to strengthen bones and reduce the risks of fractures by 30-79%. The risk of subsequent fracture is particularly high for approximately 2 years after the first fracture [5]. Identifying and treating patients early is an important step toward tackling the downstream effects of osteoporosis.
Once people at high risk are identified, a raft of often simple, low-cost interventions can contribute to improved bone health and reduce fracture risk.
After an initial fracture, the risk of subsequent fracture is particularly high for approximately the next 2 years.
= Hip Fracture
⏺ Forearm Fracture
▶ Spine Fracture
‒ Humerus Fracture
Integrate International Fragility Policy:
Strong strategic leadership in policy development is vital to ensure long-term accountability and investment. Prioritizing secondary fracture prevention will allow PFC coordination programs, such as FLS, to manage the current and future demands on the healthcare system.
Establish Comprehensive Registries and Audits:
The creation and curation of high-quality data on subsequent fragility fractures will allow accurate benchmarking of performance and pave the way for feedback and improvement at both local and national levels.
Best-Practice Models:
The Capture the Fracture® (CTF) Best Practice Framework and other models of best-practice are needed globally to increase access to high-quality post-discharge care. This will improve long-term patient outcomes.
Adequate Reimbursement Structures:
Requisite reimbursement must be mobilized to ensure access to best-practice care throughout the patient journey. This should be considered against the backdrop of the costs ensuing from failure to prevent subsequent fragility fractures within the healthcare system.
Implementing Sustainable PFC Coordination Programs
Policymakers should ensure the implementation of best-practice models in hospital care for fracture patients so that osteoporotic patients can quickly regain their independence and mobility. Among patients with hip fractures, up to 10% are likely to die while in hospital, and only half will regain the same function that they had before the fracture [6]. This can, in part, be remedied through the implementation of best-practice in-hospital care [7].
CTF’s best-practice framework supports the need for PFC coordination programs, such as FLS, implementation by defining the quality standards to prevent subsequent fractures. Our program also offers a mentorship program and virtual webinars to support the development and sustainability of PFC coordination programs, such as FLS, at the local level.
CTF has also developed robust and comprehensive Resource Center that supports PFC coordination programs to deliver on their expectations. This provides confidence to policy makers that the implemented services have a framework to local adaptation to allow delivery of patient benefit in an effective and efficient manner. It is based on learning from over 500 services in 47 countries across the globe. Link to resource center
Consistent collaboration between primary care, orthopaedics, rheumatologists, geriatrics and other services is vital to ensure that implemented services deliver the expected benefits [1,8]. Timely surgery and coordinated treatment plans led by orthogeriatricians have been shown to significantly reduce the risk of death in the short- and long-term and the likelihood of complications and prolonged hospital stays [7].
Quality Data on Secondary Fracture Identification
Policymakers require quality data on secondary fracture identification and prevention which they can use to plan and assess services. These data, however, tend not to be comprehensive, comparable or evenly spread within countries or across the globe. Additionally, most countries do not collect data on all types of fracture [9] with the majority focusing on hip fractures, meaning that vertebral or forearm fractures remain under-reported [10,11].
Regular clinical audit can act as a driver to rapidly improve secondary fracture prevention [10]. This has been amply demonstrated as a result of regular hip fracture audits in the UK and Spain [12-17]. Introducing such audits for secondary fracture prevention (encompassing all fracture types) could result in similar improvements, as observed in a comparison between China and the UK [18].
Across the globe, there is great variation in terms of how data on secondary fracture prevention is collected and analysed. This limits the ability of policymakers to compare performance between countries. In order to homogenize the approach, recent initiatives have developed standard indicators including the IOF Capture the Fracture Best Practice Framework and the Fragility Fracture Network (FFN) Minimum Common Dataset which have been adopted by several countries [12,1].
Reimbursement for diagnosis of osteoporosis is often lacking or restricted [19]. DXA scanning is a key step to diagnosing osteoporosis and identifying those at substantial risk of fractures [9]. However, reimbursement for the use of DXA remains a challenge in many countries [11]. With governments prioritizing osteoporosis as a healthcare priority, PFC coordination programs, such as FLS, will become for affordable and accessible for patients.
Automated methods of fracture identification on routine medical imaging should be the subject of further research. Deep learning and computer vision are areas of great interest and are having some success in the automated identification of vertebral fractures on CT scans [20-22].
1. Akesson K, Marsh D, Mitchell PJ et al. (2013) Capture the Fracture: a Best Practice Framework and global campaign to break the fragility fracture cycle. Osteoporos Int 24 (8):2135-2152. doi:10.1007/s00198-013-2348-z
2. Majumdar SR, Lier DA, Hanley DA et al. (2017) Economic evaluation of a population-based osteoporosis intervention for outpatients with nontraumatic non-hip fractures: the “Catch a Break” 1i [type C] FLS. Osteoporosis International 28 (6):1965-1977. doi:10.1007/s00198-017-3986-3
3. Yoshimura M, Moriwaki K, Noto S et al. (2017) A model-based cost-effectiveness analysis of osteoporosis screening and treatment strategy for postmenopausal Japanese women. Osteoporosis International 28 (2):643-652. doi:10.1007/s00198-016-3782-5
4. McLellan AR, Wolowacz SE, Zimovetz EA et al. (2011) Fracture liaison services for the evaluation and management of patients with osteoporotic fracture: a cost-effectiveness evaluation based on data collected over 8 years of service provision. Osteoporos Int 22 (7):2083-2098. doi:10.1007/s00198-011-1534-0
5. Kanis JA, Johansson H, Odén A et al. (2018) Characteristics of recurrent fractures. Osteoporos Int 29 (8):1747-1757. doi:10.1007/s00198- 018-4502-0
6. Tarazona-Santabalbina FJ, Belenguer-Varea Á, Rovira E et al. (2016) Orthogeriatric care: improving patient outcomes. Clin Interv Aging 11:843-856. doi:10.2147/cia.s72436
7. Lems WF, Dreinhöfer KE, Bischoff-Ferrari H et al. (2017) EULAR/EFORT recommendations for management of patients older than 50 years with a fragility fracture and prevention of subsequent fractures. 76 (5):802-810. doi:10.1136/annrheumdis-2016-210289
8. Javaid MK, Kyer C, Mitchell PJ et al. (2015) Effective secondary fracture prevention: implementation of a global benchmarking of clinical quality using the IOF Capture the Fracture® Best Practice Framework tool. Osteoporos Int 26 (11):2573-2578. doi:10.1007/s00198-015-3192-0
9. Kanis JA, Borgstrom F, Compston J et al. (2013) SCOPE: a scorecard for osteoporosis in Europe. Arch Osteoporos 8:144. doi:10.1007/ s11657-013-0144-1
10. ohansen A, Golding D, Brent L et al. (2017) Using national hip fracture registries and audit databases to develop an international perspective. Injury 48 (10):2174-2179. doi:10.1016/j.injury.2017.08.001
11. Hernlund E, Svedbom A, Ivergard M et al. (2013) Osteoporosis in the European Union: medical management, epidemiology and economic burden. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch Osteoporos 8:136. doi:10.1007/s11657-013-0136-1
12. Ojeda-Thies C, Sáez-López P, Currie CT et al. (2019) Spanish National Hip Fracture Registry (RNFC): analysis of its first annual report and international comparison with other established registries. Osteoporos Int 30 (6):1243-1254. doi:10.1007/s00198-019-04939-2
13. Patel NK, Sarraf KM, Joseph S et al. (2013) Implementing the National Hip Fracture Database: An audit of care. Injury 44 (12):1934-1939. doi:10.1016/j.injury.2013.04.012
14. Ferguson KB, Halai M, Winter A et al. (2016) National audits of hip fractures: Are yearly audits required? Injury 47 (2):439-443. doi:10.1016/j. injury.2015.11.018
15. Neuburger J, Currie C, Wakeman R et al. (2015) The impact of a national clinician-led audit initiative on care and mortality after hip fracture in England: an external evaluation using time trends in non-audit data. Med Care 53 (8):686-691. doi:10.1097/mlr.0000000000000383
16. . Royal College of Physicians. Fracture Liaison Service Database annual report. Benchmarking FLS improvement and performance in 2019: Pre-COVID. London: RCP, 2021.
17. International Osteoporosis Foundation, National Osteoporosis Foundation, Fragility Fracture Network. A patient-level key performance indicator set to measure the effectiveness of fracture liaison services and guide quality improvement: a position paper of the IOF Capture the Fracture Working Group, National Osteoporosis Foundation and Fragility Fracture Network. Javaid MK, Sami A, Lems W, Mitchell P, Thomas T, Singer A, Speerin R, Fujita M, Pierroz DD, Akesson K, Halbout P, Ferrari S, Cooper C. Osteoporos Int. 2020 Jul;31(7):1193-1204. PMID: 32266437; PMCID: PMC7280347.
18. Tian M, Gong X, Rath S et al. (2016) Management of hip fractures in older people in Beijing: a retrospective audit and comparison with evidence-based guidelines and practice in the UK. Osteoporos Int 27 (2):677-681. doi:10.1007/s00198-015-3261-4
19. Harvey NC, McCloskey EV, Mitchell PJ et al. (2017) Mind the (treatment) gap: a global perspective on current and future strategies for prevention of fragility fractures. Osteoporos Int 28 (5):1507-1529. doi:10.1007/s00198-016-3894-y
20. Tomita N, Cheung YY, Hassanpour S (2018) Deep neural networks for automatic detection of osteoporotic vertebral fractures on CT scans. Comput Biol Med 98:8-15. doi:10.1016/j.compbiomed.2018.05.011
21. Al-Helo S, Alomari RS, Ghosh S et al. (2013) Compression fracture diagnosis in lumbar: a clinical CAD system. Int J Comput Assist Radiol Surg 8 (3):461-469. doi:10.1007/s11548-012-0796-0
22. Lessmann N, van Ginneken B, de Jong PA et al. (2019) Iterative fully convolutional neural networks for automatic vertebra segmentation and identification. Med Image Anal 53:142-155. doi:10.1016/j.media.2019.02.005